Tissue plasminogen activator worsens experimental autoimmune encephalomyelitis by complementary actions on lymphoid and myeloid cell responses
Le 09 Avr 2021
Auteur : Pauline Hélie, Celia Camacho-Toledano, Diego Clemente, Fabian Docagne, Olivier Toutirais
Background: Tissue plasminogen activator (tPA) is a serine protease involved in fibrinolysis. It is released by
endothelial cells, but also expressed by neurons and glial cells in the central nervous system (CNS). Interestingly,
this enzyme also contributes to pathological processes in the CNS such as neuroinflammation by activating
microglia and increasing blood–brain barrier permeability. Nevertheless, its role in the control of adaptive and
innate immune response remains poorly understood.
Methods: tPA effects on myeloid and lymphoid cell response were studied in vivo in the mouse model of multiple
sclerosis experimental autoimmune encephalomyelitis and in vitro in splenocytes.
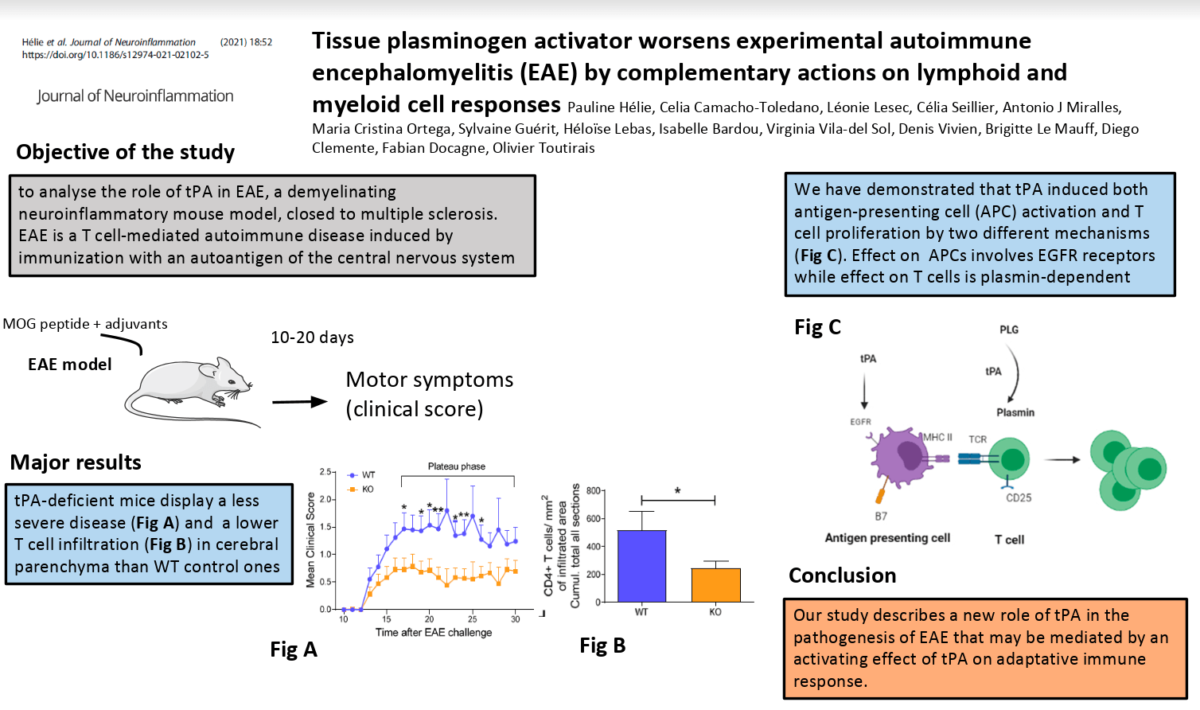
Results: tPA-/- animals exhibited less severe experimental autoimmune encephalomyelitis than their wild-type
counterparts. This was accompanied by a reduction in both lymphoid and myeloid cell populations in the spinal
cord parenchyma. In parallel, tPA increased T cell activation and proliferation, as well as cytokine production by a
protease-dependent mechanism and via plasmin generation. In addition, tPA directly raised the expression of MHCII
and the co-stimulatory molecules CD80 and CD86 at the surface of dendritic cells and macrophages by a direct
action dependent of the activation of epidermal growth factor receptor.
Conclusions
Our study provides new insights into the mechanisms responsible for the harmful functions of tPA in multiple sclerosis and its animal models: tPA promotes the proliferation and activation of both lymphoid and myeloid populations by distinct, though complementary, mechanisms.
To know more :